INFRASTRUCTURE
MANUFACTURING
SuperCure Biotech operates GMP-certified manufacturing facilities in Kundli, adhering to the latest Schedule M guidelines. These facilities are strategically located to support companies throughout the product development and commercialization process.
Our manufacturing facilities are meticulously designed to meet GMP standards, with dedicated quality control and assurance units ensuring the highest manufacturing quality. We specialize in healthcare services, including the production of raw materials such as our trademarked product, Supercell, a trusted name in core excipients for pharmaceutical needs. With over 30 years of experience, we also manufacture pellets, particularly focusing on gastro-related products, making it the largest facility of its kind in North India.
In addition to raw materials, we offer contract manufacturing services, guiding products from packaging to commercialization using our own high-quality materials, ensuring both quality and cost-effectiveness. Our extensive manufacturing capacity encompasses various dosage forms, supported by cutting-edge technology and specialized expertise in producing specialty formulations.
Supported by a seasoned team of professionals, we provide a diverse range of products and state-of-the-art facilities, offering flexible and cost-effective manufacturing solutions with value-added services. Our formulation range includes tablets, capsules, ready-mix coatings, core excipients, pellets, granules, Ayurvedic syrups, and protein powders.
Our Manufacturing facility is build with the state of art technology which meets cGMP requirements of Schedule-M, ICH, WHO, EDQM etc.,”
We do have advanced manufacturing equipments, with different capacity Fluid Bed Coaters , Conventional coating pans, Sifters, Tray driers, Pulverizers, Blenders, Fully automatic capsule filling ,Fluid Bed dryers ,State of the art packaging equipment for best quality finishes etc., all made up of SS material.
Our Manufacturing facility is supported with best Air Handling Systems which are supported with HEPA filters and Air Conditioning to maintain clean room environment and to provide ambient climatic zone to support manufacturing process and Healthy Environment for work force, which meets ISO Class 8 clean room requirements.
Water used in the Process is generated by best in class Dual RO – EDI water system.



CAPACITY
-
Dosage FormAnnual Capacity
-
TABLETS75 cr
-
CAPSULES60 cr
-
PELLETS360 Ton
-
READY MIX COATING600 Ton
-
PROTEIN9 Lakh Jars
| Dosage Form | Annual Capacity |
|---|---|
| TABLETS | 75 cr |
| CAPSULES | 60 cr |
| PELLETS | 360 Ton |
| READY MIX COATING | 600 Ton |
| PROTEIN | 9 Lakh Jars |
-
Dosage FormAnnual Capacity
-
TABLETS75 cr
-
CAPSULES60 cr
-
PELLETS360 Ton
-
READY MIX COATING600 Ton
-
PROTEIN9 Lakh Jars
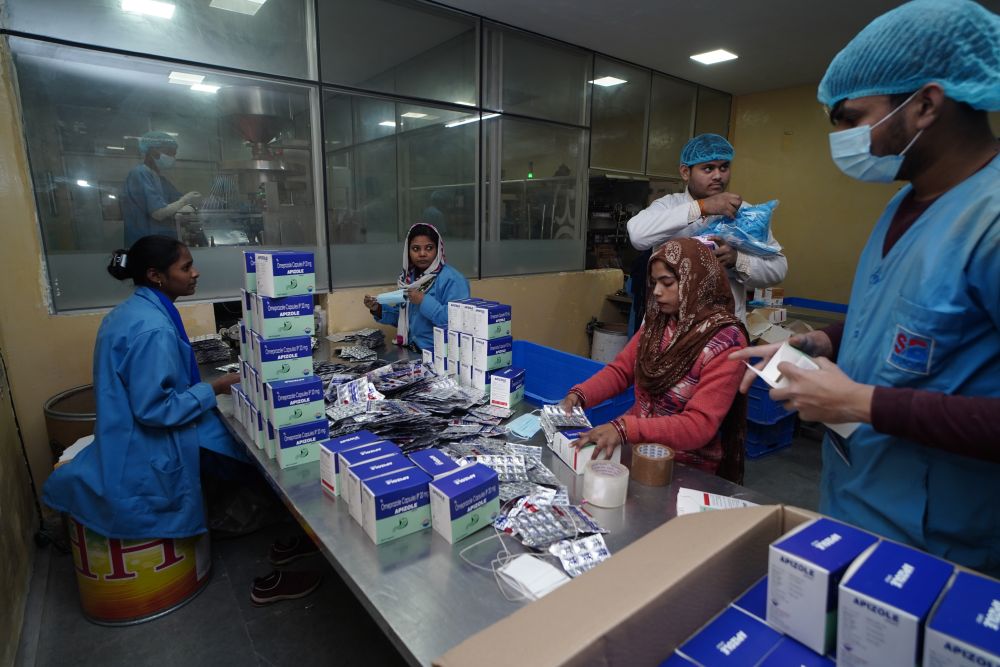
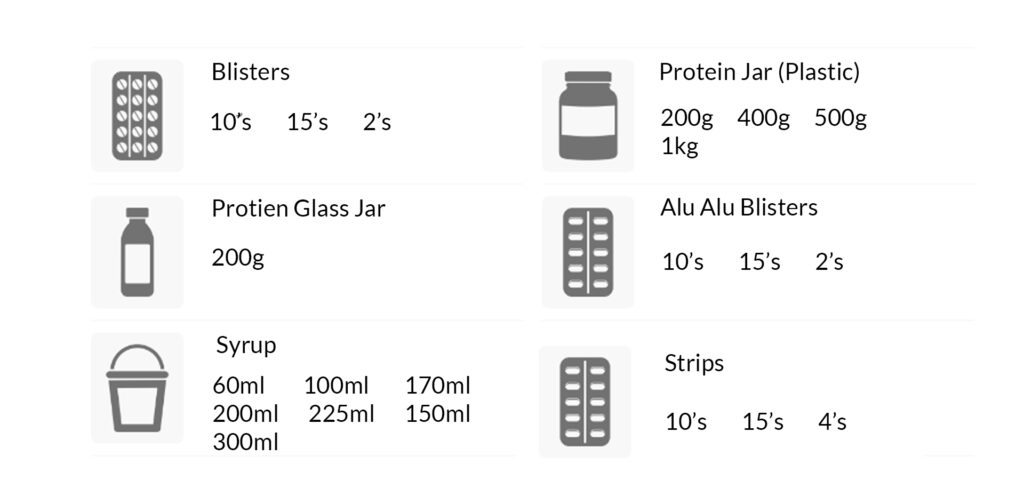
PACKAGING









OUR QUALITY MANAGEMENT SYSTEM
QUALITY ASSURANCE
QUALITY CONTROL

QC Department Comprises:
- Air Conditioned QC Laboratory with Analytical Instruments
- Documentation Meeting Regulatory Standards
- Control Samples and Stability Chamber
- Analytical Method Validation
- Quality Audits and Investigations
- Documentation and Record System
Every batch undergoes comprehensive quality checks, documented in Batch Manufacturing Records. Finished products are analyzed inline with in-house and pharmacopeia standards, with details recorded for further distribution.
Quality Control Department Comprises:
- Air Conditioned QC Laboratory with Analytical Instruments
- Documentation Meeting Regulatory Standards
- Control Samples and Stability Chamber
- Analytical Method Validation
- Quality Audits and Investigations
- Documentation and Record System:
- Every batch undergoes comprehensive quality checks, documented in Batch Manufacturing Records. Finished products are analyzed inline with in-house and pharmacopeia standards, with details recorded for further distribution.









Our Quality Policy:
At Supercure Biotech Pvt Ltd, we are committed to :
Customer Satisfaction through Quality Products
Compliance with Legal and Regulatory Requirements
Continuous Improvement of Quality Management Systems
Integration of Quality, Environment, Health & Safety in all Processes.

